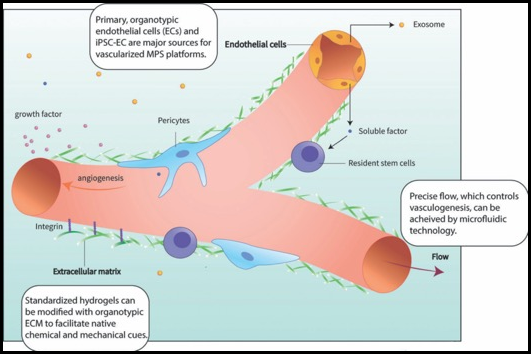
In recent years, microphysiological system (MPS, also known as, organ-on-a-chip or tissue chip) platforms have emerged with great promise to improve the predictive capacity of preclinical modeling thereby reducing the high attrition rates when drugs move into trials. While their designs can vary quite significantly, in general MPS are bioengineered in vitro microenvironments that recapitulate key functional units of human organs, and that have broad applications in human physiology, pathophysiology, and clinical pharmacology. A critical next step in the evolution of MPS devices is the widespread incorporation of functional vasculature within tissues. The vasculature itself is a major organ that carries nutrients, immune cells, signaling molecules and therapeutics to all other organs. It also plays critical roles in inducing and maintaining tissue identity through expression of angiocrine factors, and in providing tissue-specific milieus (i.e., the vascular niche) that can support the survival and function of stem cells. Thus, organs are patterned, maintained and supported by the vasculature, which in turn receives signals that drive tissue specific gene expression. In this review, we will discuss published vascularized MPS platforms and present considerations for next-generation devices looking to incorporate this critical constituent. Finally, we will highlight the organ-patterning processes governed by the vasculature, and how the incorporation of a vascular niche within MPS platforms will establish a unique opportunity to study stem cell development.
The vascular niche in next generation microphysiological systems